Description
Specially Engineered Device
For High Flow Rate Applications
Optimized 0.2 µm Supor® membrane achieves 23% higher flow rates for shorter infusion times.
- 100% bacterial retention* provides added safety and patient protection.
- High capacity dual vents for faster repriming times and orientation-independent venting.
- Same materials of construction as IV-3 filter simplify validation requirements.
- Maximizes delivery of critical drugs with proven low drug binding Supor membrane to increase dosing accuracy.
* 0.2 µm Supor membrane is retentive of B. diminuta and meets USP 25/NF20 requirements for a sterilizing grade filter per ASTM F838-83 test methods.
Specifications
Materials of Construction
Media: Supor (polyethersulfone) membrane
Housing: Modified acrylic
Vent: 0.02 µm supported PTFE
Bubble Point
46 psi
Minimum Water Flow Rate
> 40 mL/min @ 91 cm (36 in.) head pressure
Maximum Operating Pressure
45 psi
Pyrogenicity
< 0.25 EU/mL using the LAL test method
Sterilization Compatibility
EtO, Gamma irradiation (tested to 50 kGy)
Biological Safety
Materials of contruction pass USP Biological Reactivity Tests, In Vivo <88>
Applications
- Apheresis
- Drug delivery
- IV therapy
Performance
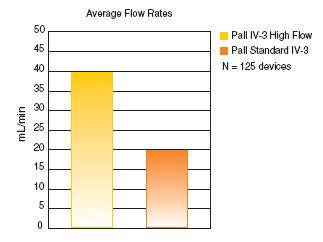
Results of internal evaluations show that the IV-3 High Flow filter delivers flow rate performance that is 23% better than the traditional IV-3 filter design.
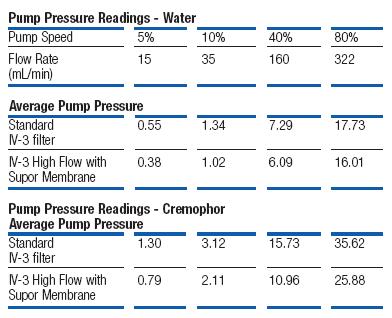
In high flow delivery applications, pumps are used to generate the flow rates required. The IV-3 High Flow filter was tested against Pall's traditional IV-3 filter design by comparing pump pressure required to maintain various flow rates using water and a cremophor solution. The results are shown below.
Type
Use
Ordering Information
| Part Number | Description | Packaging |
| 6494521 | IV-3 High Flow with Supor membrane, clear/green housing | 2000/pkg |
Reviews
Earn 10% off* your next order online by leaving a review of this product. Please login to your account to leave a review. We appreciate and value your feedback.
*Subject to Terms and Conditions.